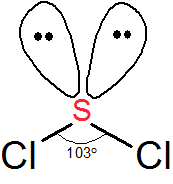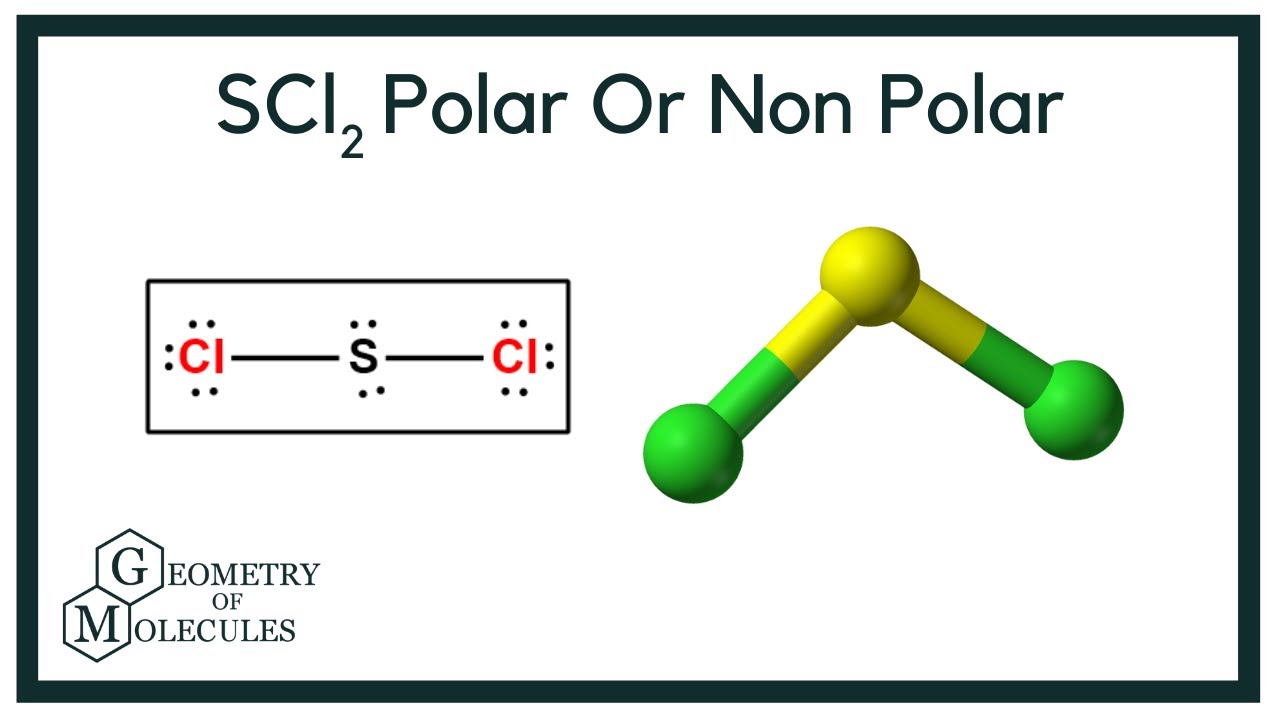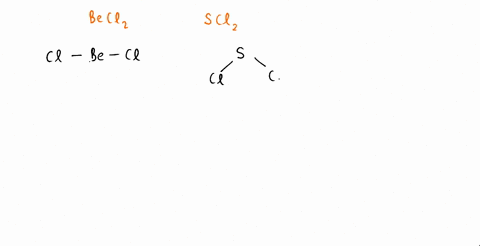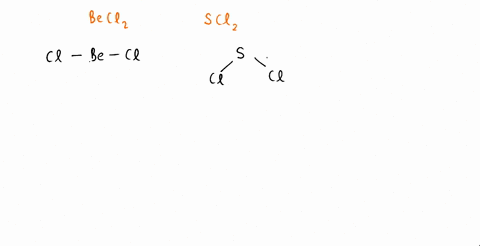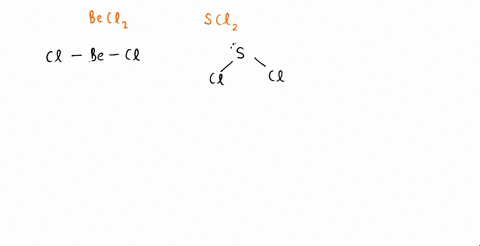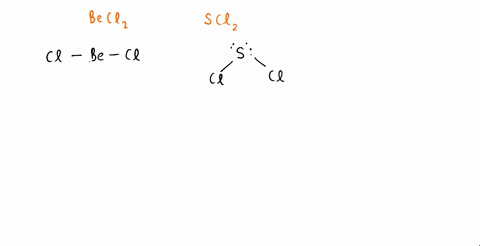
SOLVED: An SCl2 molecule has (polar or nonpolar) bonds and a (bent, linear, tetrahedral, trigonal planar, trigonal pyramidal) molecular shape. As a result, an SCl2 molecule is .(polar or nonpolar)

The molecules CO2 and SCl2 are both of the general molecular type AB2, but one is polar and one is not. Which molecule is polar? Draw the Lewis structures for these molecules

what is the direction and how do you find it for the dipole moment in scl2? to the 2 or the cl? | Homework.Study.com
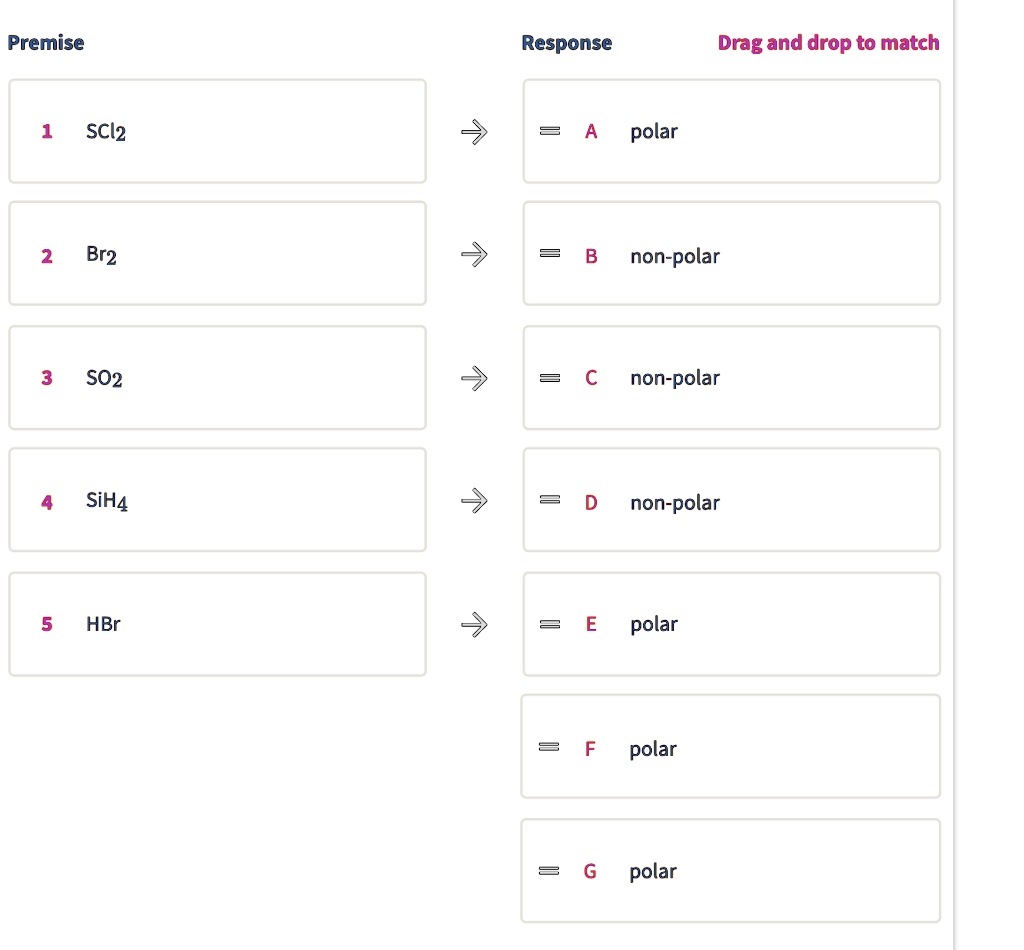
SOLVED: Premise Response Drag and drop to match polar Br2 non-polar S02 non-polar SiHA non-polar HBr polar polar SCl2 polar

Why is SCl2 polar?I have the Lewis structure and from there I can see that it has polar bonds because of the Cl so both S - Cl bonds are polar bonds.