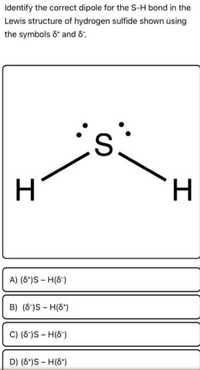
Use formal charges to identify the better Lewis structure. a) Structure I is the better Lewis structure. b) Structure II is the better Lewis structure. c) Neither structure is better than the

Effect of External Electric Field on Tetrel Bonding Interactions in (FTF3···FH) Complexes (T = C, Si, Ge, and Sn) | ACS Omega
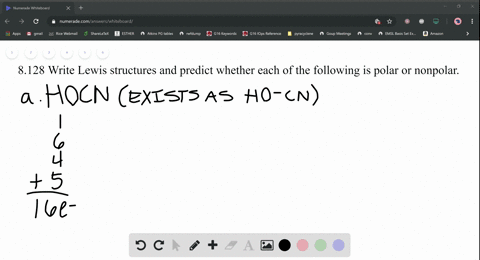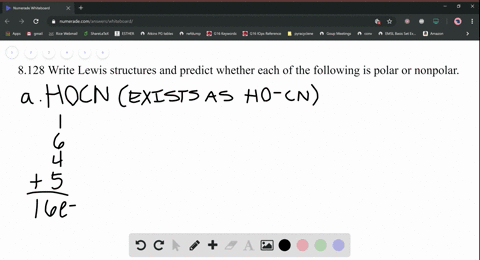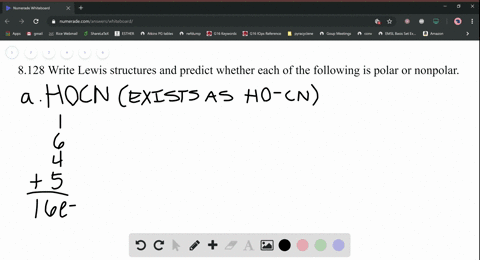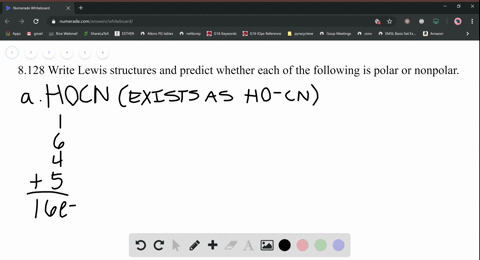
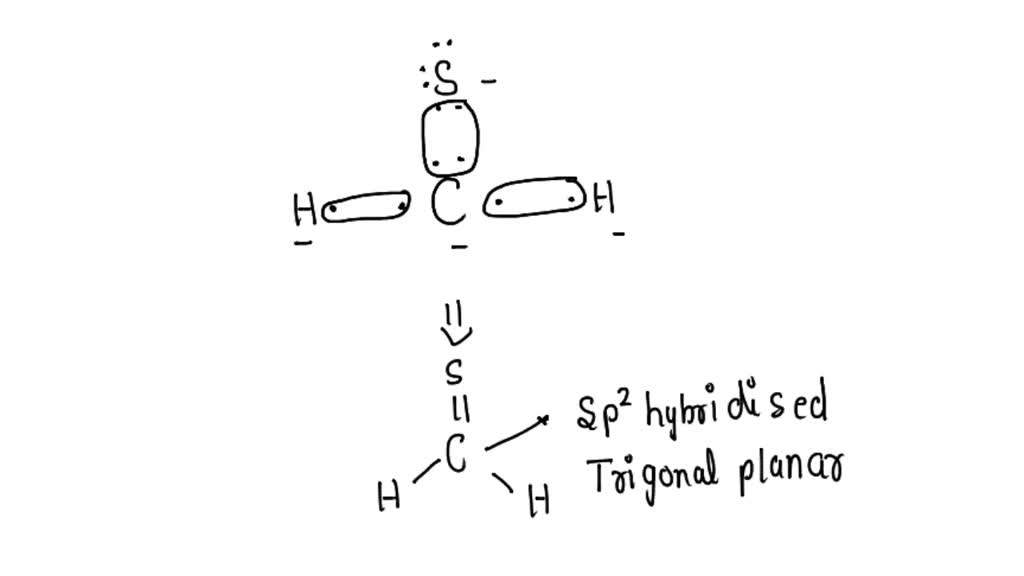
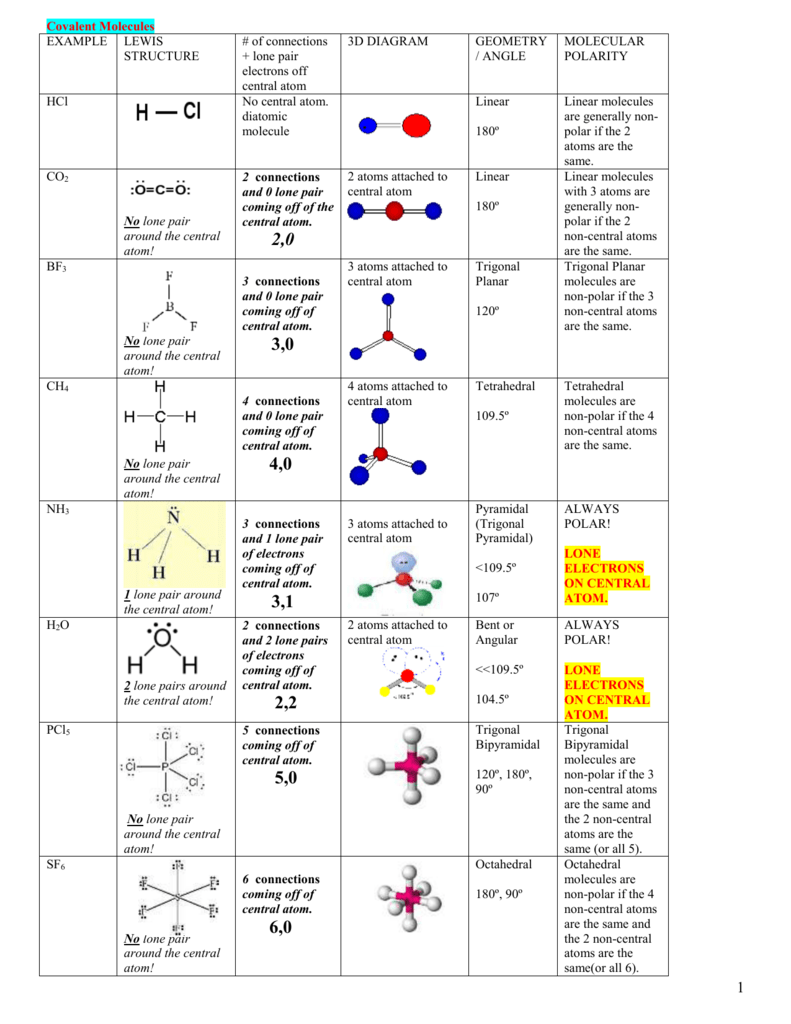
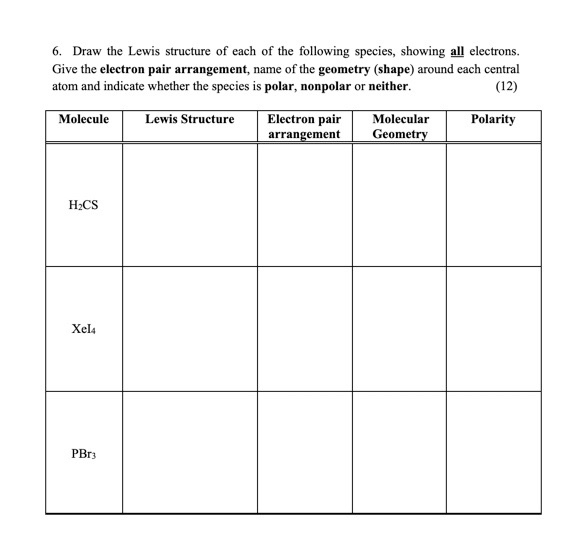
SOLVED: What is the Lewis structure for H2CS, with the central atom of C. Is the molecular polar or nonpolar? Identify the intermolecular forces present?
Liquids | Free Full-Text | Revision and Extension of a Generally Applicable Group Additivity Method for the Calculation of the Refractivity and Polarizability of Organic Molecules at 298.15 K